The wastewater treatment microbiome is made up of free-floating microorganisms and biofilm or flocculated microorganisms, most of which are bacteria and some archaea. The surrounding environments that these microorganisms are in contact with is the hydrosphere and atmosphere (provided via aeration). For how the microbiome interacts with the surrounding environment, the example of partial nitrification in wastewater is discussed below.
Partial Nitrification
Biological nitrogen removal in wastewater can be achieved via combination of partial nitrification and Anammox. This process does not require external addition of carbon, it also produces a negligible amount of sludge and requires less energy and oxygen (Ge et al., 2015). Anammox requires the accumulation of nitrite which is achieved by partial nitrification.
The diagrams below show the chemical pathways by which ammonium and ammonia. AMO refers to the enzyme ammonia monooxygenase, HAO refers to the enzyme hydroxylamine oxidoreductase, AOB refers to ammonium oxidising bacteria and NOB refers to nitrite oxidising bacteria.
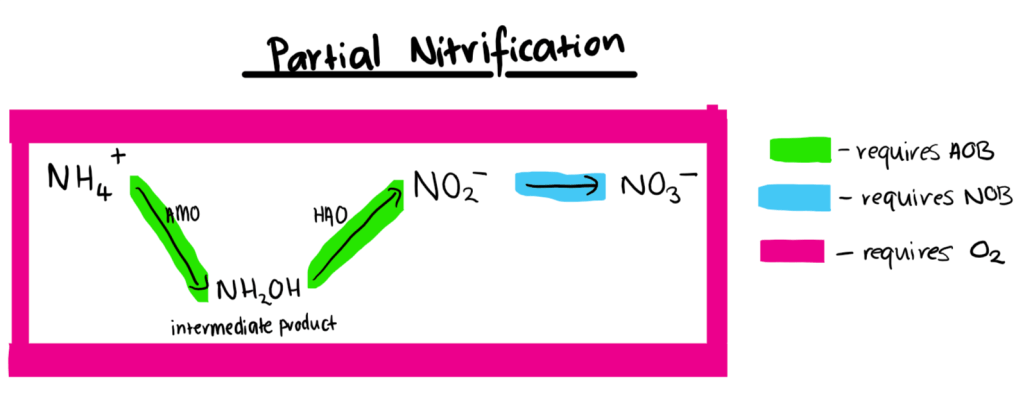
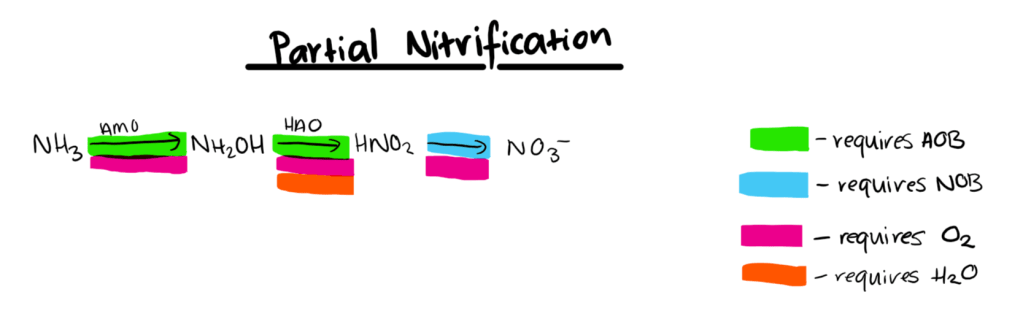
Interactions with Environment
As ammonium oxidation to nitrite is the limiting step of the nitrification pathway, the main strategy used to achieve partial nitrification is to accumulate AOB and inhibit or washout NOB (Ge et al., 2015). By inhibiting NOB, nitrite is available for anammox bacteria to utilise and convert to nitrogen gas, thus completing the nitrogen removal process.
AOB have a lower oxygen half saturation constant than NOB, thus allowing for enrichment at lower levels of dissolved oxygen (DO) (Ge et al., 2015). By toggling DO levels, nitrite accumulation and ammonia oxidation levels will change, in addition to other microbial processes. For example, at low DO concentration, nitrite accumulation still occurs, but filamentous bulking may occur within the sludge (Ge et al., 2015), often this process causes issues with settleability of bioflocs.
Real time control using ammonium–nitrogen probes can be used to determine the endpoint of nitrification in an automatic feed-forward system (Ge et al. 2015), thus making it possible to keep an optimal DO level for partial nitrification. When aeration is stopped, it allows for the accumulation of nitrite. In the absence of oxygen, NOB cannot oxidise nitrite to nitrate, where anammox bacteria can complete the nitrogen cycle (convert nitrite to nitrogen gas) without requiring the presence of NOB and denitrifiers. NOB typically have lower growth rates when subjected to anoxic conditions whilst AOB do not exhibit any impact (Ge et al., 2015).
pH also influences the process of partial nitrification, this is via free ammonia (FA) and free nitrous acid (FNA) concentration changes, which can be used to control the activity of AOB and NOB. A pH of 7.5-8.5 enhances nitrite accumulation and much higher levels of FA inhibit AOB than opposed to the levels required to inhibit NOB (Ge et al., 2015). Thus, it is possible to inhibit NOB without impacting AOB at low FA levels. FNA interferes with the transmembrane pH gradient required for ATP synthesis by donating a proton (Ge et al., 2015). As NOB are also more sensitive to AOB, it is possible to run a plant that inhibits NOB and not AOB at FNA levels below 0.42 mg N/L (Ge et al. (2015).
Other factors such as substrate concentration and temperature also affect the growth and metabolic processes of AOB and NOB.
References
Ge, S., Wang, S., Yang, X., Qiu, S., Li, B., Peng, Y. (2015). Detection of nitrifiers and evaluation of partial nitrification for wastewater treatment: A review. Chemosphere, 140, 85-98. doi: 10.1016/j.chemosphere.2015.02.004